Potentiostats / Galvanostats
An advanced potentiostat and galvanostat is a high-precision electrochemical instrument that precisely controls and measures voltage (potentiostat mode) or current (galvanostat mode) between electrodes in an electrochemical cell. It enables the detailed study and analysis of electrochemical reactions, which are crucial for research in corrosion, battery development, fuel cells, sensors, and electrocatalysis. These instruments are often modular and scalable, offering versatile functions l ike impedance spectroscopy, cyclic voltammetry, and charge-discharge testing to support cutting-edge scientific and industrial applications. The combined potentiostat/galvanostat functionality allows seamless switching between voltage-controlled and current-controlled experiments, enhancing flexibility and accuracy in electrochemical research and development.
Portable Potentiostat

Single Channel

Bipotentiostats & RRDE Systems

Multichannel

Modular

Current Boosters

Specialised Devices and Modules
Potentiostats & Galvanostats for Electrochemical Research
Potentiostats and galvanostats are essential electrochemical instruments widely used in scientific and industrial research to control and measure electrical parameters within electrochemical systems. These instruments are often combined into one platform. This makes it easy to switch between two modes. You can choose potentiostatic mode, which is voltage-controlled, or galvanostatic mode, which is current-controlled. The choice depends on the goal of the experiment. This dual functionality is critical for applications such as corrosion testing, battery development, electrocatalysis, and sensor fabrication.
ScienceGears proudly offers a variety of potentiostats and galvanostats that researchers and industries in Australia and New Zealand trust.
We have systems for various fields. It doesn't matter if you are working on:
- Battery testing
- Corrosion studies
- Electrocatalysis
- Biosensors
- Photoelectrochemistry
We can support your needs. We offer everything from basic models to advanced research-grade platforms.
We provide expert help, quick support, and fast delivery from leading global brands. These include Zahner Elektrik, Admiral Instruments (Squidstat series), and Corrtest Instruments.
What Is a Potentiostat and How Does It Work?
A potentiostat is a precise tool. It controls the voltage between a working electrode and a reference electrode. At the same time, it measures the current between the working electrode and a counter electrode. In electrochemical research, it is essential for exploring redox reactions, studying electrode interfaces, and developing advanced energy storage systems.
Potentiostats are commonly used in conjunction with techniques like cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), chronoamperometry, and more.
Potentiostat vs Galvanostat: Understanding the Difference
A potentiostat controls voltage and measures current, while a galvanostat controls current and measures voltage. Many modern instruments are dual-mode systems, offering both functionalities. At ScienceGears, we stock multiple models with switchable modes for added flexibility in your experiments.
Applications of Potentiostats
- Batteries & Supercapacitors – Charge/discharge profiling, cycle life testing
- Corrosion Science – Polarisation curves, EIS, Tafel plots
- Fuel Cells & Electrolysers – Impedance, power output, electrocatalyst development
- Biosensors – Low current detection using screen-printed electrodes
- Photoelectrochemistry – With light sources for solar-driven reactions
Need help selecting the right model for your application? Contact our team for personalised support.
Featured Potentiostat Models
Explore our curated range of potentiostats below or view our single-channel models for entry-level and budget-conscious labs.
Product Families and Features
Squidstat Potentiostats / Galvanostats
User-friendly, modern instruments designed for flexible electrochemical testing with strong value propositions for academic and industrial users.
M7 Modular Potentiostats / Galvanostats High-performance modular platforms developed with decades of expertise in electrochemical impedance spectroscopy (EIS), allowing for advanced configuration and customisation.
- IM7x Next-generation modular potentiostat / galvanostat
- IM7 Modular Potentiostat/Galvanostat
- IM7C Compact potentiostat / galvanostat
External Power Potentiostats
Four-quadrant external potentiostats extend the current and voltage capabilities of Zennium PRO or Zennium X, enabling high-demand experiments.
Optional Addon Cards / Modules
Accessory modules are designed to expand functionality for advanced applications in kinetic modelling and complex system simulation.
CorrTest Single Channel Potentiostats / Galvanostats
High-speed digital platforms delivering robust data acquisition, stable output, and accurate analysis in corrosion science and fundamental electrochemistry.
- CS350M
- CS310M
- CS300M
- CS100E (with EIS) – Portable potentiostat
- CS100 (without EIS) – Portable potentiostat
CorrTest BiPotentiostats / BiGalvanostats
CorrTest bipotentiostat and bigalvanostat are advanced electrochemical instruments designed for dual-channel and high-current applications. The bipotentiostats enable simultaneous control of two working electrodes, ideal for electrocatalysis, redox comparisons, and EIS-based sensor studies. Bigalvanostats deliver stable, multi-channel current control for battery testing, electroplating, and fuel cell evaluation in high-demand research environments.
CorrTest Multichannel Potentiostats / Galvanostats
Multi-channel configurations for parallel testing, ideal for battery arrays, corrosion screening, and multi-electrode experimentation.
CorrTest Current Boosters and Specialized Devices
High-current booster modules and specialized systems for pitting corrosion analysis and electrochemical mapping.
- 20A/40A/100A Current Booster
- CS300M for pitting corrosion
- CST520 Multi-electrode Array Electrochemical Mapper
Summary
ScienceGears offers robust potentiostat and galvanostat solutions for a wide spectrum of electrochemical applications. The Squidstat, Zennium, and CorrTest series offer a range of systems. They include compact benchtop units and modular high-performance options. These systems give researchers precise control and reproducibility. They also allow for easy integration in many fields. These fields include corrosion engineering, battery research, fuel cell development, and electrocatalysis. Other areas are electroplating, supercapacitor testing, water splitting, biosensing, and electrochemical impedance spectroscopy (EIS). Our platforms use advanced software and flexible configurations. They also have special expansion modules. These features help support modern scientific workflows. They are built for long-term reliability and flexibility.
FAQs
1. What is a Potentiostat
A potentiostat is an electronic device used in electrochemical research. It controls the voltage between a working electrode and a reference electrode. At the same time, it measures the current flowing to or from the working electrode. It plays a central role in techniques like cyclic voltammetry, chronoamperometry, and electrochemical impedance spectroscopy (EIS).
Potentiostats are essential for studying redox reactions, energy storage devices, corrosion, electrocatalysis and electrochemical sensors. By keeping a steady potential and measuring the current, researchers can study electron transfer, diffusion, and material properties at the electrode-electrolyte interface.
It uses three electrodes:
Working Electrode (WE): Where the chemical reaction happens.
Reference Electrode (RE): A stable point to compare voltage.
Counter Electrode (CE): Completes the circuit.
2. How does a potentiostat work?
A potentiostat is an electronic device used in electrochemistry. It controls and maintains the voltage of a working electrode. This is done in relation to a stable reference electrode. The device also measures the current flowing through the system.
Here’s a clear and simplified explanation of its working principle:
Key Components of a Potentiostat:
A typical potentiostat involves a three-electrode system:
- Working Electrode (WE): The electrode at which the electrochemical reaction of interest occurs.
- Reference Electrode (RE): An electrode with a stable and known potential, used as a fixed reference point.
- Counter Electrode (CE) / Auxiliary Electrode: Completes the circuit by balancing the current passing through the working electrode.
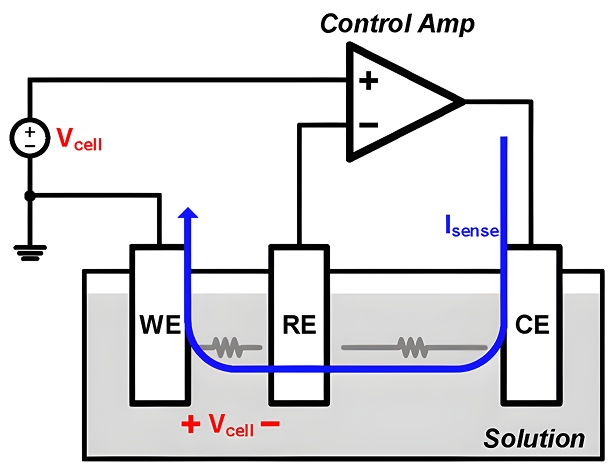
Working Principle (Step-by-Step):
Potential Control
- The potentiostat applies a controlled voltage between the Working Electrode and the Reference Electrode.
- The Reference Electrode provides a stable, known reference potential, ensuring accurate control and measurement.
Current Measurement
- Once the set potential is applied, the electrochemical reaction occurs at the Working Electrode, causing electrons to flow.
- This electron flow (current) moves through the electrolyte and completes the circuit via the Counter Electrode.
- The potentiostat precisely measures the current resulting from this reaction.
Feedback Control
- The potentiostat uses an operational amplifier (op-amp) and feedback loops. It keeps the desired potential at the Working Electrode. It continually adjusts as the reaction happens.
- This feedback mechanism rapidly compensates for changes in the reaction environment, ensuring constant potential.
3. What is the difference between a potentiostat and a galvanostat?
The main difference is in the control variable. A potentiostat controls the voltage and measures the current response. A galvanostat controls the current and measures the voltage.
Potentiostatic control is often used to study reaction rates and mechanisms. Galvanostatic control is commonly used for battery charging and discharging, electroplating, and measuring corrosion rates. Many modern instruments can switch between these modes to suit different experimental needs.
Potentiostat vs. Galvanostat: Key Differences
|
Feature / Aspect |
Potentiostat |
Galvanostat |
|
Primary Control |
Voltage (potential) |
Current |
|
Measured Output |
Current |
Voltage |
|
Control Mechanism |
Maintains constant voltage between WE and RE |
Maintains constant current through the cell |
|
Main Use Cases |
Redox studies, corrosion, EIS, CV, electrochemical sensors |
Battery cycling, electroplating, corrosion under fixed current |
|
Electrode Setup |
Three-electrode system (WE, RE, CE) |
Three-electrode system (WE, RE, CE) |
|
Response to Cell Changes |
Adjusts the current to maintain voltage |
Adjusts voltage to maintain current |
|
Example Techniques |
Cyclic voltammetry, chronoamperometry, EIS |
Galvanostatic charge/discharge, LSV with constant current |
|
Common in |
Research labs, sensor development, surface chemistry |
Battery labs, corrosion testing, material stress studies |
|
Feedback Loop Goal |
Keep WE at the desired potential |
Keep current through the WE constant |
|
When to Use |
When understanding electrochemical reaction mechanisms |
When applying a specific current load is critical |
4. What parameters should I consider when buying a potentiostat?
Key specifications include compliance voltage, maximum current, current resolution, EIS frequency range, and number of channels. Your application (e.g., batteries, corrosion, sensors) also plays a significant role.
5. Can I use ScienceGears potentiostats with screen-printed electrodes?
Yes, we offer several models. These include options from Squidstat and CardTest. They work with screen-printed electrodes and have cable adapters for easy use.
6. What is a floating mode potentiostat?
A floating mode potentiostat is a device that works without being connected to the earth ground. This means its internal parts are electrically separate from the main ground. This lets the potentiostat safely measure electrochemical systems that are not grounded. This includes batteries in series, fuel cell stacks, solar panels, or cells in metallic autoclaves.
Using floating mode removes the risk of ground loops. Ground loops can create noise, lead to wrong data, or even harm the equipment. Ground loops occur when multiple paths to ground create unintended current flow through sensitive measurement circuits.
Floating potentiostats are very helpful in hydrogen permeation experiments. In these tests, the electrochemical cell is often inside a metal pressure vessel. It can also be linked to high-impedance gas detection systems. In such setups, direct grounding could lead to short circuits or interfere with signal integrity. The floating configuration allows accurate measurement of transient hydrogen diffusion signals without interference from the metallic housing or other grounded lab equipment.
Other common use cases include:
- Testing individual cells within high-voltage battery stacks
- Electrochemical monitoring inside metal reactors
- Electrochemistry on grounded substrates (e.g., metal sheets or pipelines)
In short, a floating potentiostat provides electrical safety, signal stability, and versatility in complex or grounded environments.
7. What is a bipotentiostat?
A bipotentiostat is a special device. It can control two working electrodes separately. Both electrodes use the same reference and counter electrode. It is commonly used in studies involving dual-electrode systems, such as generator-collector configurations, electrochemical sensors, or RRDE experiments.
This enables the simultaneous monitoring of two related reactions, allowing for mechanistic studies with high temporal resolution.
8. What is the purpose of a reference electrode?
The reference electrode gives a stable, known voltage. This voltage is used to measure and control the working electrode's voltage. Unlike the counter electrode, it does not carry current, ensuring that the potential remains constant throughout the experiment.
Common reference electrodes are Ag/AgCl, saturated calomel electrode (SCE), and Hg/HgO. Each one is chosen based on the solvent and the needs of the experiment. A stable reference is critical for reproducibility and accurate interpretation of electrochemical results.
9. What is the function of a counter electrode?
The counter electrode, or auxiliary electrode, helps complete the circuit. It allows current to flow through the electrochemical cell. It is usually made from materials like platinum or carbon. These materials do not take part in the important electrochemical reaction.
Its main job is to balance the current going through the working electrode. This helps the potentiostat keep the right potential at the working electrode.
10. How do I choose the right potentiostat?
To pick the right potentiostat, consider a few key factors. First, look at the voltage range. Next, check the current range. Also, think about the EIS capability. Don't forget to consider the number of channels and the sampling rate. These factors should match your specific application. For example, battery and corrosion studies may need higher currents. In contrast, sensor research may focus on low noise and sensitivity.
Also evaluate software usability, data export formats, and instrument expandability. If you plan to scale your research, opt for modular or multi-channel models.
11. Is a built-in EIS necessary?
If your work involves impedance characterisation of batteries, coatings, sensors, or fuel cells, built-in EIS is highly beneficial. It enables frequency sweeps from mHz to MHz without external modules.
For users not working with impedance, EIS may be optional, but is often a worthwhile future-proof feature.
12. What is a dummy cell?
A dummy cell is a test circuit composed of resistors, capacitors, or diodes that simulate predictable electrochemical behaviour. It’s used to validate potentiostat performance, calibrate EIS, and troubleshoot noise or instability.
Dummy cells are essential tools for training and diagnostics.
13. Do you provide training and support?
Yes. We offer installation, remote/onsite training, and full after-sales support across Australia and New Zealand.
Need Help Choosing the Right Potentiostat?
Our team can help you find the best instrument for your research needs. Whether you're working with fuel cells, biosensors, solar cells, or corrosion testing — we've got you covered.
How To Choose The Right Potentiostat
Request Expert Advice
