1 Introduction: The Gap Between Making a Battery and Understanding One
Building a battery electrode is one problem. Understanding what is happening inside it during charge and discharge is an entirely different one — and it is the second problem that separates research groups producing publishable, reproducible data from those generating results they cannot explain.
The potentiostat/galvanostat is one of the key instruments that closes that gap. It allows researchers to apply a controlled electrochemical stimulus — a defined voltage, current, or perturbation — and record the response with the resolution and accuracy needed to extract meaningful physical parameters from real materials.
This guide covers the complete electrochemical characterisation workflow for battery research: how to connect a potentiostat to a battery correctly for half-cell and full-cell configurations, what the Galvanostatic Intermittent Titration Technique (GITT) and Potentiostatic Intermittent Titration Technique (PITT) actually measure and why they matter, and which cell testing protocols produce data that holds up under scrutiny — from internal group meetings through to peer review.
Across Australian battery research — including lithium-ion, sodium-ion, solid-state and flow-battery work — these techniques are widely used to connect electrochemical behaviour with material performance. Getting the measurement protocol right is essential for reproducible, defensible results.
2 Why Battery Research Needs a Potentiostat Not Just a Cycler
Before getting into connection protocols, it is worth being precise about what a potentiostat does that a battery cycler does not — because the distinction shapes every decision about which instrument to use at which stage of your research.
A battery cycler applies current and measures voltage. It is designed for long-duration cycling — thousands of charge/discharge cycles — with high channel count and robust, reliable operation. It tells you capacity, coulombic efficiency, and cycle life. It is essential for ageing studies and rate performance characterisation.
A potentiostat/galvanostat controls voltage or current with measurement capability that is often better suited to diagnostic electrochemistry, depending on the model, current range, voltage range and system configuration. It can:
- Apply potential steps to the millivolt level and measure current responses to the microampere level
- Sweep potential at precisely controlled scan rates
- Perturb the system with small-amplitude sinusoidal signals across a wide frequency range, depending on the instrument model and EIS configuration
- Extract physical parameters — diffusion coefficients, charge transfer resistances, double layer capacitances — that a standard battery cycler without diagnostic electrochemical capability may not access
The question is never “cycler or potentiostat.” It is “what does this stage of the research actually need to measure?” In most battery research programmes, the answer is both — and knowing when to use each is what separates efficient research from expensive data collection.
People Also Ask: What is the difference between a battery cycler and a potentiostat for battery research?
A battery cycler applies current and measures voltage, optimised for long-duration cycling of many cells simultaneously — delivering capacity, coulombic efficiency, and cycle life data. A potentiostat controls voltage or current and, depending on the model and options fitted, may support techniques such as EIS, cyclic voltammetry, GITT and PITT — enabling extraction of physical parameters such as diffusion coefficients and charge-transfer resistance that a cycler cannot measure. Most serious battery research programmes use both: cyclers for ageing and rate testing, potentiostats for diagnostic characterisation and mechanistic analysis.
3 How to Connect a Potentiostat to a Battery: Configuration and Protocol
Half-Cell Configuration The Starting Point for Most Battery Research
The vast majority of electrode material characterisation in battery research uses a half-cell configuration: a two-electrode arrangement in which the material under study (the working electrode) is paired against a lithium metal counter/reference electrode in a relevant non-aqueous electrolyte.
This is how to connect a potentiostat to a battery in the half-cell format.
Cell formats and their connection approach:
- Coin cell (CR2032 or CR2016): A common format for academic battery research. For lithium-metal and moisture-sensitive non-aqueous cells, assemble inside a suitable inert-atmosphere glovebox, with H₂O and O₂ levels appropriate to the electrolyte and cell chemistry. Once sealed, connect as follows: working electrode (WE) lead → electrode under study; reference electrode (RE) and counter electrode (CE) leads joined together → lithium metal counter/reference electrode. Do not rely only on coin-cell can polarity; confirm the cell orientation and electrode placement before connection.
For two-electrode configurations, many potentiostats either require the RE and CE leads to be shorted together or provide a dedicated two-electrode mode. Always follow the instrument manual for the correct lead arrangement. On selected Squidstat and Zahner configurations available through ScienceGears, two-electrode operation can be configured through the relevant software or hardware setup, depending on the model. - Swagelok-style T-cell: Preferred when a true three-electrode configuration is needed — for example, when independently monitoring electrode potentials in a full cell, or when using a lithium reference wire. The T-cell’s third port accommodates a lithium metal reference wire connected to the RE terminal of the potentiostat, while WE and CE connect to the electrode under study and counter electrode, respectively, according to the experiment design.
- Pouch cell: For pouch cells, tab material and polarity depend on the electrode design. In a two-electrode half-cell, connect the WE lead to the electrode under study and connect the CE/RE leads to the lithium metal counter/reference electrode. Ensure tab connections are clean, tightly clamped, and making full electrical contact — a resistive tab connection introduces a contact resistance artefact that can be misinterpreted as electrode impedance in EIS measurements.
Verifying the Connection Before Testing
Before running any electrochemical protocol, perform these three checks:
- Measure Open Circuit Voltage (OCV) immediately after connection. Compare against the expected value for the cell’s state of charge. A freshly assembled lithium-ion half-cell against lithium metal should show an OCV consistent with the electrode chemistry, state of charge and cell design. A cathode half-cell, an anode half-cell and a full cell will have different expected OCV ranges. An OCV outside this range may indicate a short circuit, reversed polarity, electrolyte leakage, an incorrect connection or an unexpected cell condition.
- Monitor OCV stability for 5–10 minutes. Rapid OCV drift (> ±5 mV per minute) in a freshly assembled cell indicates an active self-discharge mechanism, electrolyte decomposition at a reactive electrode surface, or a micro-short through the separator. Resolve before testing.
- Run a single slow CV scan (e.g., 0.1 mV s⁻¹) across the full potential window before any GITT, PITT, or rate test. This identifies the major redox features of your electrode material, confirms the potential window is appropriate, and establishes whether any unexpected reactions are occurring.
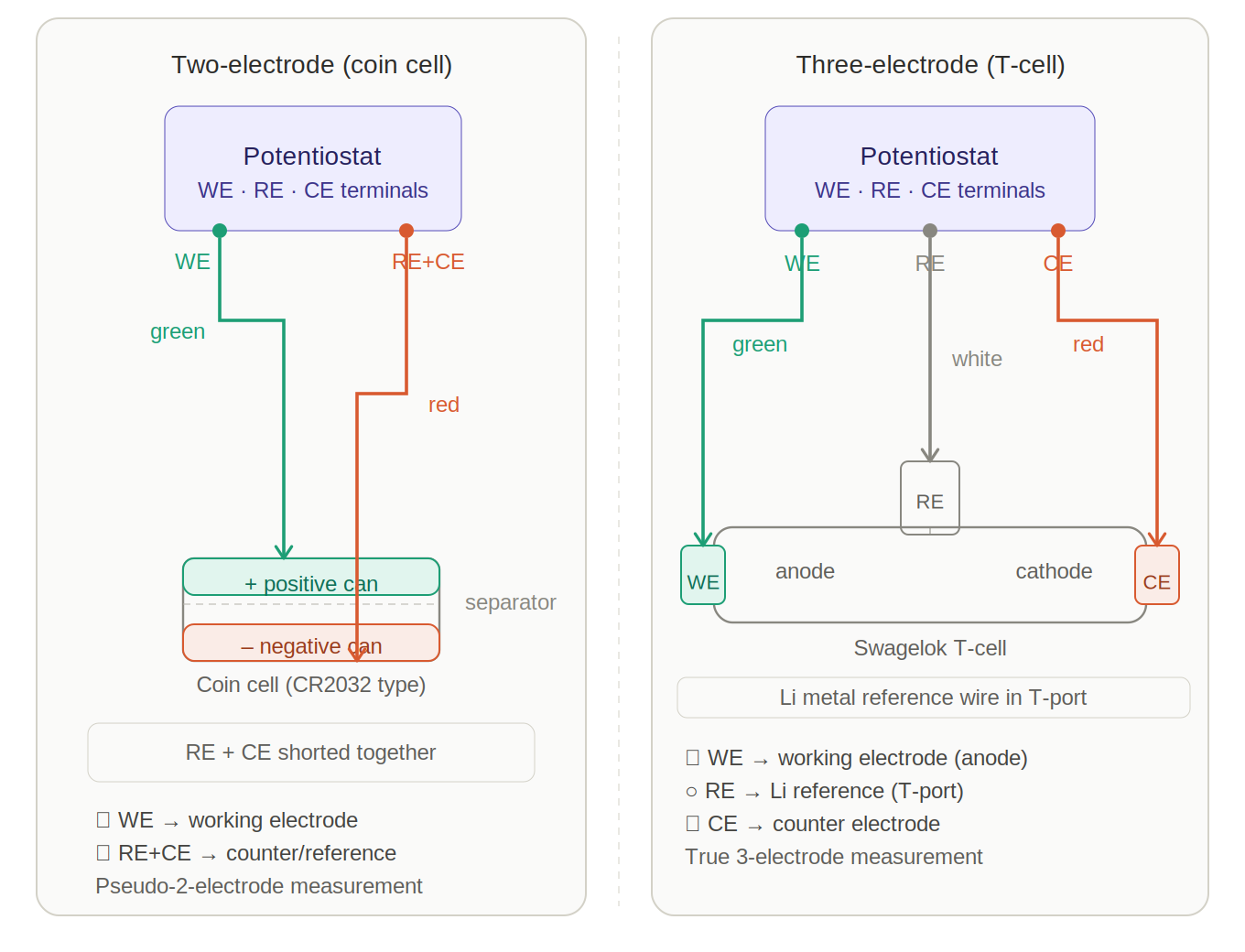
Left panel — coin cell (two-electrode): The WE lead connects to the terminal connected to the electrode under study; the RE and CE leads are shorted together and connected to the lithium metal counter/reference terminal. This pseudo-two-electrode arrangement is commonly used for galvanostatic testing of coin cells, but the cell orientation must always be confirmed before connection.
Right panel — Swagelok-style T-cell (three-electrode): Each port is independent — WE connects to the electrode under study, CE connects to the counter electrode, and RE connects to the lithium reference electrode in the third port. This enables true three-electrode measurements and is useful for GITT, EIS, and CV characterisation when independent electrode-potential monitoring is required.
The colour coding is illustrative only. Always follow the potentiostat manufacturer’s cable labels, manual, and software configuration for the correct WE, CE, RE, and sense-lead connections.
4 GITT: Measuring What Actually Limits Your Battery
What GITT Is and Why It Matters
The Galvanostatic Intermittent Titration Technique (GITT) answers a question that every battery electrode material researcher eventually confronts: how fast can lithium (or sodium, or potassium) ions actually move through my electrode material — and how does that mobility change as the electrode charges and discharges?
That question matters because solid-state ion diffusion is frequently the rate-limiting step in battery performance. An electrode material with outstanding thermodynamic properties — high capacity, ideal redox potential — can still fail to deliver that capacity at practical charge rates if lithium ions cannot diffuse through it fast enough. GITT makes that diffusion kinetics problem quantitative.
The GITT Protocol: Step by Step
The GITT protocol alternates short galvanostatic (constant current) pulses with extended open-circuit relaxation periods:
- Current pulse phase: Apply a constant current for a defined duration — typically 10 to 30 minutes — at a low rate (commonly C/10 to C/20). The current drives lithium ions into or out of the electrode, shifting its potential. The magnitude of the transient voltage response during the pulse reflects both the thermodynamic and kinetic properties of the material at that state of lithiation.
- Relaxation phase: Turn the current off. The cell rests at open circuit whilst the lithium concentration gradient established during the pulse equilibrates by diffusion. The time required for the potential to reach a new steady-state — which can range from 30 minutes to 4 hours per step depending on the material’s diffusivity — directly reflects how slowly or quickly lithium ions redistribute within the electrode particles.
- Extracting the diffusion coefficient: For the commonly used short-pulse approximation, the apparent lithium-ion diffusion coefficient can be estimated as:
D_Li ≈ (4 / πτ) × (m_B V_M / M_B S)² × (ΔE_s / ΔE_t)²
where τ is the current-pulse duration, m_B is the active-material mass, V_M is the molar volume, M_B is the molar mass, S is the electroactive area, ΔE_s is the steady-state voltage change after relaxation, and ΔE_t is the transient voltage change during the current pulse, usually after excluding the IR drop. This expression gives an apparent diffusion coefficient and is valid only when the short-pulse GITT assumptions are reasonable.
Repeating this across the full charge/discharge range — typically every 5–10% state of charge — maps how diffusivity changes with lithiation state. This map is essential data for understanding why certain materials perform well at low C-rates but fail at high rates, and for guiding structural engineering of electrode particles to improve rate capability
People Also Ask: What does GITT measure in battery research?
GITT — the Galvanostatic Intermittent Titration Technique — measures the chemical diffusion coefficient of lithium (or sodium) ions through a battery electrode material as a function of state of charge. A short constant-current pulse is applied, followed by an open-circuit relaxation period. From the ratio of the transient voltage response during the pulse to the steady-state voltage step, the diffusion coefficient is calculated at each point across the charge/discharge range. This data reveals whether solid-state ion diffusion is the rate-limiting step in the electrode’s performance and guides materials engineering to improve fast-charging capability.
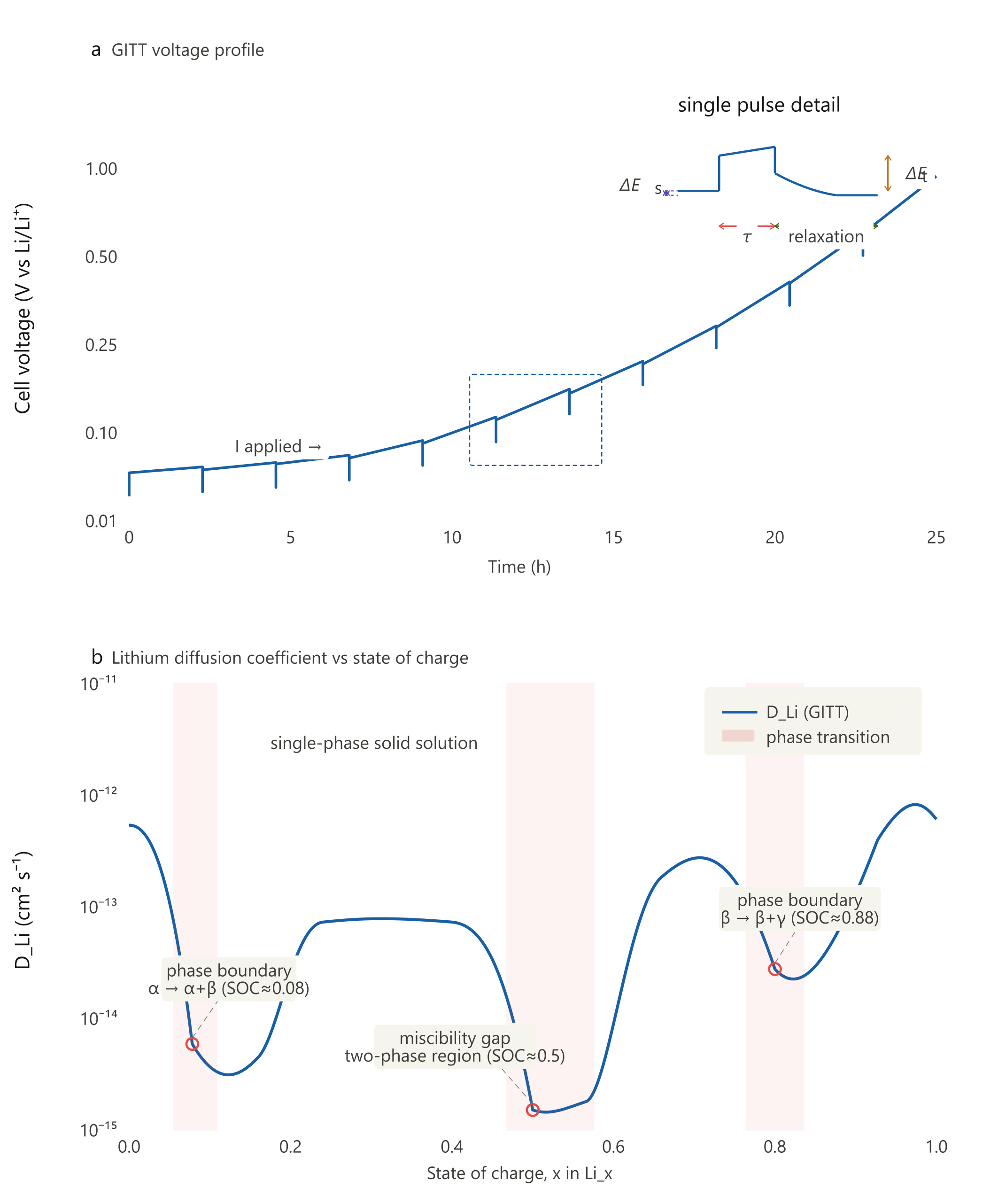
The annotated GITT two-panel figure illustrates the key features commonly examined during GITT analysis:
Panel A — Voltage Profile
The sawtooth waveform shows the repeating pulse-relaxation morphology across a full half-cell charge. A single GITT transient labels all four key parameters: τ (current pulse duration), the relaxation window, ΔEₜ (instantaneous voltage jump at pulse onset), and ΔEₛ (net equilibrium step between successive relaxed baselines).
Panel B — DLi vs SOC: The diffusivity curve shows an illustrative log-scale variation across lithiation. In this example, annotated minima mark possible phase-transition or transport-limited regions (highlighted in red bands):
- SOC ≈ 0.08 — the α → α+β two-phase boundary (e.g., dilute → phase-coexistence onset)
- SOC ≈ 0.50 — the deep trough at the miscibility gap centre where ionic transport is most restricted
- SOC ≈ 0.88 — a secondary minimum at the β → β+γ boundary near full lithiation
For the commonly used short-pulse GITT approximation, the apparent lithium-ion diffusion coefficient can be estimated as:
D_Li ≈ (4 / πτ) × (m_B V_M / M_B S)² × (ΔE_s / ΔE_t)²
where τ is the current-pulse duration, m_B is the active-material mass, V_M is the molar volume of the active material, M_B is the molar mass, S is the electrode/electroactive area, ΔE_s is the steady-state voltage change after relaxation, and ΔE_t is the transient voltage change during the current pulse after excluding the iR drop
5 PITT: The Potentiostatic Counterpart
What PITT Measures and When to Use It
The Potentiostatic Intermittent Titration Technique (PITT) is the potentiostatic complement to GITT. Where GITT applies a current and measures the voltage response, PITT applies a voltage step and measures the current decay — and from that current transient, extracts the same fundamental quantity: the chemical diffusion coefficient of ions through the electrode material.
The choice between GITT and PITT is not arbitrary. It depends on the thermodynamic characteristics of your electrode material:
- Use GITT when: The electrode material has a sloping, featureless voltage profile across the composition range (e.g., graphite in the disordered region, many oxide cathodes); you want diffusivity values at defined states of charge expressed as composition fractions; or current control is more reliable than voltage control for your specific cell configuration.
- Use PITT when: The material has a steep, sharply featured voltage profile where small composition changes produce large voltage changes — making current pulses difficult to control precisely; phase transition materials (e.g., LiFePO₄, which undergoes a two-phase reaction with a flat voltage plateau) where the voltage resolution of PITT offers advantages; or you want to combine diffusion coefficient extraction with thermodynamic data from the quasi-equilibrium current-composition relationship.
The PITT Protocol
The potentiostat steps the cell potential by a small increment — typically in the millivolt range, depending on the material and protocol — and holds that potential constant whilst recording the current as a function of time. The current decay may include double-layer charging, interfacial processes, and diffusion-controlled behaviour. In suitable cases, the diffusion-controlled region can be analysed using Cottrell-type or finite-diffusion models.
For a simplified semi-infinite diffusion approximation, the current decay is commonly related to t⁻¹⁄². For battery electrodes, the appropriate fitting model should be selected based on particle geometry, phase behaviour, and boundary conditions.
From the diffusion-controlled region of the current transient, an apparent diffusion coefficient may be estimated when the model assumptions are valid. The process can be repeated at each voltage step across the selected potential window.
PITT experiments are inherently slower than GITT — each potential step must be held until the current decays to a defined fraction of its initial value (typically <1% of the peak current), which can take hours per step for materials with low diffusivity. However, for materials where voltage resolution is the critical parameter — particularly phase-transition cathode materials — PITT provides data that GITT cannot cleanly deliver.
People Also Ask: What is the potentiostatic intermittent titration technique (PITT) and how does it differ from GITT?
PITT applies small voltage steps to a battery electrode and measures the resulting current decay over time, extracting the lithium diffusion coefficient from the Cottrell equation applied to the diffusion-controlled current tail. GITT instead applies current pulses and measures voltage transients. PITT can be useful for phase-transition electrode materials with steep or flat voltage profiles where precise potential control provides additional insight, although interpretation should consider phase-boundary movement and model limitations. Both techniques measure solid-state diffusion coefficients but approach the measurement from opposite electrochemical domains — PITT from the potentiostatic side, GITT from the galvanostatic side.
6 Standard Battery Cell Testing Protocols
Beyond GITT and PITT, the standard battery characterisation workflow with a potentiostat/galvanostat covers four essential protocol types. These are the experiments that contextualise the diffusion data, establish the electrochemical identity of the material, and build the comparative dataset that makes GITT and PITT results interpretable.
Cyclic Voltammetry (CV) — Fingerprinting the Material
Run CV at slow scan rates (0.01–0.5 mV s⁻¹) across the full potential window before any other characterisation. The CV provides the electrochemical fingerprint of your electrode material — every oxidation and reduction peak corresponds to a specific lithium insertion/extraction event, phase transition, or side reaction.
Key diagnostic information from the CV:
- Peak positions — confirm the expected redox chemistry and detect unexpected reactions
- Peak separation (ΔEp) — indicates kinetic reversibility; large separation suggests slow solid-state diffusion or high ohmic resistance
- Peak current scaling with scan rate — distinguishes diffusion-controlled (battery-like) from capacitive (supercapacitor-like) charge storage mechanisms
- Cycle-to-cycle evolution — identifies activation behaviour in first cycles, SEI formation, and early-stage degradation
Electrochemical Impedance Spectroscopy (EIS) — Seeing Inside the Cell
EIS is a powerful diagnostic technique in battery research. By perturbing the cell with a small-amplitude sinusoidal signal — commonly 5–10 mV, depending on cell stability and instrument configuration — across a selected frequency range, EIS can help separate the total cell impedance into physical contributions when interpreted with an appropriate equivalent-circuit or physics-based model.
A well-measured and appropriately fitted EIS spectrum for a lithium-ion half-cell may resolve:
- RΩ (ohmic resistance) — electrolyte resistance plus contact resistances; manifests as the high-frequency real-axis intercept in the Nyquist plot
- RSEI and CSEI — resistance and capacitance of the solid electrolyte interphase layer; appears as a high-to-mid frequency semicircle
- Rct and Cdl — charge-transfer resistance and double-layer capacitance at the electrode-electrolyte interface; mid-frequency semicircle
- W (Warburg impedance) — solid-state diffusion limitation; the characteristic 45° line in the low-frequency Nyquist region
Tracking how these components evolve with state of charge and cycle number provides mechanistic insight into degradation that capacity curves alone cannot deliver. An electrode that loses 20% capacity over 100 cycles tells you that degradation is occurring. EIS measured at selected cycle intervals can help identify likely contributing processes and interfaces when combined with suitable modelling and complementary measurements.
Selected ScienceGears NEWARE EIS-capable battery cycler configurations integrate impedance measurement into cycling protocols, reducing the need to manually transfer cells between instruments for periodic EIS measurements during long-term ageing studies.
Rate Capability Testing — Quantifying Power Performance
Rate capability testing charges and discharges the cell at progressively higher C-rates — typically C/10, C/5, C/2, 1C, 2C, 5C, and 10C — with a defined number of cycles at each rate and a recovery check at C/10 after the high-rate sequence.
The capacity retention across the rate sequence quantifies the material’s power capability: how much of the theoretical capacity remains accessible as charge rate increases. A material that retains 95% capacity at 1C but only 60% at 5C shows a rate limitation that may involve solid-state diffusion, charge-transfer resistance, ionic/electronic transport, or contact resistance. GITT, EIS, and complementary testing can help identify the dominant limitation and guide materials engineering strategies such as particle-size control, surface coating, or structural modification.
Calendar and Cycle Ageing
For research groups studying battery degradation — increasingly relevant as Australia’s energy storage sector scales — a combined battery cycler and potentiostat/galvanostat workflow enables systematic ageing and diagnostic protocols:
- Calendar ageing holds cells at defined temperature and state of charge for weeks or months, with periodic diagnostic interruptions (CV + EIS + capacity check) to monitor degradation rate. An increase in Rct from EIS can be a sensitive indicator of interfacial degradation, although it should be interpreted alongside capacity data, voltage profiles, and complementary characterisation.
- Cycle ageing applies a defined charge/discharge protocol — often constant current/constant voltage (CC-CV), depending on the cell chemistry — for extended cycling, with periodic EIS diagnostics to track changes in SEI resistance, interfacial impedance, loss of active lithium, and possible signatures associated with lithium plating on graphite anodes.
7 The ScienceGears Battery Research Ecosystem
Battery research at the electrode material level — GITT, PITT, CV, EIS — and battery research at the cell production and ageing level — cycling, calendar ageing, rate testing — require different instruments. Most equipment suppliers treat these as separate conversations. ScienceGears treats them as one.
The reason is straightforward: the researchers asking about potentiostat protocols for GITT are often the same researchers who will need a battery cycler for long-term ageing studies six months later. And the researchers setting up a cell production workflow for coin cells or pouch cells are often building towards a characterisation programme that requires potentiostat-level diagnostic capability. Fragmenting those needs across multiple suppliers means losing the thread that connects the electrochemical measurement to the material decision to the cell design choice.
ScienceGears’ battery research portfolio spans the complete workflow:
Electrochemical Characterisation Instruments
- Potentiostat/galvanostat systems (Squidstat, Zahner, Corrtest) — single-channel, multichannel, bipotentiostat, and modular configurations
- NEWARE Battery Cycler Series — CT-4000 through CT-9000, including the EIS Battery Cycler Series with integrated impedance measurement
- Battery Environmental Test Chambers — temperature-controlled environments for calendar and cycle ageing studies
Battery Test Cells and Hardware
- Coin cell hardware (CR2032, CR2016, CR2025 formats)
- Swagelok T-cells and custom three-electrode configurations
- Pouch cell and supercapacitor assembly fixtures
- In-situ and operando electrochemical cells for synchrotron and Raman studies
Battery Materials
- Cathode materials (layered oxides, phosphates, spinels)
- Anode materials (graphite, silicon, hard carbon, lithium metal)
- Electrolytes, salts, separators, binders, conductive carbons, and current collectors
- Sodium-ion battery materials for next-generation research programmes
When Dr. Kalai Govindasamy works through a battery characterisation setup with a research group, the conversation typically covers the electrode material chemistry, the cell format, the specific electrochemical questions the group is trying to answer, and the timeline of the research programme. The instrument recommendation follows from that picture — not from what is easiest to ship.
8 Pre-Test Checklist: Battery Electrochemical Characterisation
Before connecting a potentiostat to a battery cell and running any protocol, work through this checklist:
- ☐ Cell assembled in glovebox (H₂O < 0.1 ppm, O₂ < 0.1 ppm) and rested for minimum 12 hours before testing
- ☐ OCV measured immediately after connection — within expected range for cathode chemistry and state of charge
- ☐ OCV stable for 5 minutes — drift < ±5 mV per minute before proceeding
- ☐ Potential window verified against cathode and anode stability limits — no risk of electrolyte decomposition or lithium plating
- ☐ Potentiostat current range appropriate for expected peak currents at planned C-rate
- ☐ Two-electrode vs three-electrode configuration confirmed and leads connected correctly
- ☐ Background CV run at 0.1 mV s⁻¹ to confirm electrode activity and absence of unexpected reactions
- ☐ EIS baseline recorded at OCV before first charge — reference impedance for degradation tracking
- ☐ GITT or PITT current/voltage step size confirmed against rate and relaxation time requirements
- ☐ Data save format and file naming convention set before starting long-duration experiments
9 Summary
The gap between knowing that a battery electrode material works and understanding why it works — or why it fails at high rates, or why it degrades faster than expected — is closed by electrochemical measurement. Connecting a potentiostat to a battery correctly, running GITT to map solid-state diffusion across the full charge range, applying the potentiostatic intermittent titration technique to extract diffusion coefficients from phase-transition materials, and building a complete EIS baseline before long-term cycling begins — these are core components of a well-designed battery characterisation programme.
The techniques in this guide are established methods used across battery research programmes internationally to connect electrochemical measurements with material behaviour, diffusion limitations, and degradation mechanisms.
If your battery research programme is at the electrode material characterisation stage, the cell design stage, or the ageing study stage — and if the electrochemical measurement side of that programme needs better instruments, better protocols, or both — the conversation starts the same way at ScienceGears: with your research question, not with a product list. Request a quote or get in touch directly.
Explore ScienceGears’ full battery research instrument and materials portfolio at batteries energy storage, browse our application notes for in-depth technical guides, or contact the team to discuss your characterisation requirements.
Related Articles in This Series
- ← Potentiostat Applications in Modern Electrochemical Research: A Complete Guide
- → Potentiostat for Green Hydrogen: PEM Electrolysis Conditioning and Testing Protocols
- → Potentiostat Setup: 3-Electrode Cell, Electrode Connections and Experiment Preparation
- → What Does a Potentiostat Do? Applications Across Research Sectors in AU and NZ
- → Potentiostat for Biosensors and Electrochemical Sensing: A Lab Guide






