1 What is Cyclic Voltammetry?
Cyclic voltammetry (CV) is an electrochemical technique that lets you observe how a chemical species gains and loses electrons — simply by sweeping a voltage back and forth and measuring the current that flows. It is, in essence, a real-time window into the chemistry of electron transfer.
In plain terms, you apply a voltage to an electrode sitting in a solution. As the voltage rises, some molecules in the solution start to give up electrons (they get oxidised). When you reverse the voltage, those same molecules take electrons back (they get reduced). Your instrument records the current at every voltage point, and the resulting graph called a cyclic voltammogram tells you an enormous amount about the chemistry happening at that electrode surface.
Core Definition — CV
Cyclic voltammetry is an electrochemical technique in which the voltage applied to a working electrode is swept linearly from a starting potential to an end potential, and then reversed — while a potentiostat measures the current response at each point. The resulting current vs. potential plot is called a cyclic voltammogram.
First developed in the 1950s and 1960s, CV has become one of the most widely used diagnostic techniques in electrochemistry. It is fast to set up, non-destructive in most cases, and provides rich qualitative and quantitative information in a single experiment. Whether you are studying battery materials, developing a biosensor, screening new drug molecules, or characterising a catalyst, cyclic voltammetry is almost always the first technique you reach for.
Key Insight
Cyclic voltammetry is sometimes described as the "first experiment" of electrochemistry — the technique researchers run first to understand a new system before applying more specialised measurements. If you have a new electrode material or compound, CV is where you start.
2 How Does Cyclic Voltammetry Work?
Understanding CV requires knowing two things: what a potentiostat does, and how an electrochemical cell behaves when voltage is applied. Let us walk through each step simply.
Step 1 : The Potentiostat Controls the Voltage
A potentiostat is the instrument at the heart of every CV experiment. Its job is to precisely control the voltage (potential) applied between two electrodes in your solution, and to simultaneously measure the current flowing through the cell. Depending on the instrument and settings, it can acquire data fast enough to capture the dynamic chemistry happening during the experiment.
Step 2: The Potential Sweep
When you run a CV experiment, you programme the potentiostat to start at an initial potential and ramp the voltage linearly in one direction at a fixed rate. This rate is called the scan rate (measured in millivolts per second, mV/s). When the voltage reaches your chosen end potential, the sweep reverses direction and the voltage ramps back the other way. This forward-and-reverse motion is the "cyclic" part of cyclic voltammetry.
Step 3: Redox Reactions Occur at the Electrode
As the voltage changes, it drives different reactions at the working electrode surface. When the voltage reaches the characteristic oxidation potential of a species in solution, electrons are pulled away from those molecules — and current spikes upward, creating an oxidation peak on your graph. When the voltage sweeps back through the reduction potential, electrons are given back to those molecules — and current spikes in the opposite direction, creating a reduction peak.
These two peaks — one for oxidation, one for reduction — are the signature of a reversible electrochemical reaction. Their positions, heights, and shapes tell you about the thermodynamics and kinetics of the electron transfer process.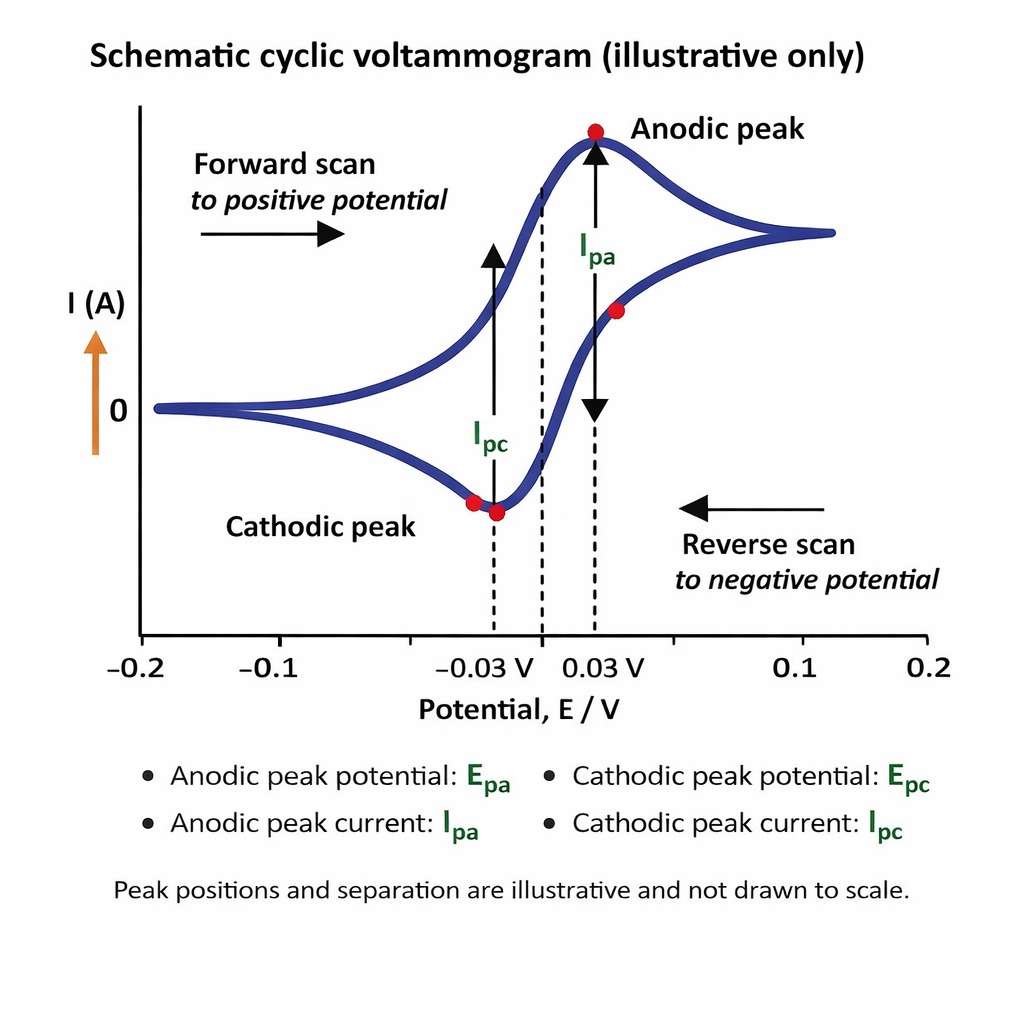
Figure 1 — Cyclic Voltammogram Diagram
Applied Potential (V) → x-axis | Current (I) → y-axis
Forward sweep (oxidation, +current): Voltage increases → oxidation peak reached at Epa (Anodic Peak)
Reverse sweep (reduction, −current): Voltage decreases → reduction peak at Epc (Cathodic Peak)
Figure 1 — A typical cyclic voltammogram. Epa marks the anodic (oxidation) peak; Epc marks the cathodic (reduction) peak. The separation between these two peaks reveals information about the reversibility and kinetics of the redox
3 Equipment You Need for Cyclic Voltammetry
Running a CV experiment requires four core components. Understanding what each does will help you set up your experiment correctly and troubleshoot problems when they arise.
Working Electrode (WE)
The surface where your electrochemical reaction of interest occurs. Common materials: glassy carbon, gold, platinum, or a modified electrode surface. This is where you focus your chemistry.
Reference Electrode (RE)
Provides a stable, known potential against which the working electrode potential is measured. Common types: Ag/AgCl (aqueous), SCE, or Ag/Ag⁺ (non-aqueous). Never pass current through it.
Counter Electrode (CE)
Completes the electrical circuit by carrying the current returning from the working electrode. Platinum wire or graphite rod are most common. The CE reaction is unimportant — only the WE chemistry matters.
| Component | What It Does | Typical Examples | Cost Indication |
|---|---|---|---|
| Potentiostat | Controls potential, measures current, records the voltammogram | Squidstat Solo, CS350M, Zennium X | AUD $1,500–$30,000+ |
| Working Electrode | Surface where redox reaction occurs | Glassy carbon, gold disc, screen-printed electrode | AUD $20–$300 |
| Reference Electrode | Provides stable potential reference | Ag/AgCl (3M KCl), SCE, Ag/Ag⁺ | AUD $50–$200 |
| Counter Electrode | Completes the circuit | Platinum wire, graphite rod | AUD $30–$150 |
| Electrochemical Cell | Contains the electrolyte solution and electrodes | 3-electrode glass cell, H-cell, flow cell | AUD $100–$500 |
| Software | Programmes the experiment, records and displays data | Supplied with potentiostat (Zahner, Admiral, CorrTest) | Included or licensed |
If you are just starting out, a three-electrode glass cell containing a simple redox couple such as potassium ferrocyanide in potassium chloride solution is an excellent first system. It produces a clean, textbook-perfect voltammogram that lets you verify your equipment and learn to interpret results before moving to your real samples.
4 How to Read a Cyclic Voltammogram
The cyclic voltammogram can look intimidating at first, but once you understand the key features, it becomes a very readable document of your chemistry. Here is what to look for.
The Oxidation Peak (Anodic Peak, Epa)
On the forward sweep, as the voltage increases, you will typically see a current peak in the positive direction. This is the anodic (oxidation) peak, labelled Epa. It occurs at the potential where your molecule of interest loses electrons most readily. The height of this peak (the peak current, Ipa) is related to how much of the molecule is present in solution and how fast it diffuses to the electrode.
The Reduction Peak (Cathodic Peak, Epc)
When the sweep reverses, you will see a current peak in the negative direction. This is the cathodic (reduction) peak, labelled Epc. It occurs when the oxidised species from the forward sweep is reduced back to its original form as the potential drops below the reduction potential.
What the Peak Separation Tells You
For a perfectly reversible, fast redox couple measured under ideal conditions, the theoretical separation between Epa and Epc is 59 mV at room temperature (per electron transferred). In practice, you will often see larger separations due to uncompensated resistance, slow electron transfer kinetics, or adsorption effects. Monitoring how the peak separation changes with scan rate is one of the classic ways to characterise the kinetics of your redox system.
Formal Potential (E°′)
The formal potential, sometimes called the half-wave potential, is calculated as the midpoint between Epa and Epc:
This value approximates the standard reduction potential of the redox couple and is one of the most important numbers you extract from a CV experiment.
The Current Ratio (Ipa / Ipc)
For a reversible reaction, the ratio of the anodic peak current to the cathodic peak current should equal 1. If this ratio deviates from 1, it suggests that the reaction has some irreversibility — perhaps a follow-up chemical reaction consumes the oxidised or reduced product before it can be converted back.
5 Understanding Scan Rate in Cyclic Voltammetry
Scan rate is one of the most important variables you control in a CV experiment, and understanding its effect is essential for correct data interpretation.
The scan rate is the speed at which the potential is swept — typically expressed in millivolts per second (mV/s) or volts per second (V/s). Common scan rates range from 5 mV/s for slow systems to 10 V/s or more for studying fast electron transfer kinetics.
The Randles–Ševčík Relationship
For a reversible, diffusion-controlled redox reaction, the peak current scales with the square root of the scan rate. This is expressed by the Randles–Ševčík equation:
Where Ip is the peak current and ν (nu) is the scan rate. If you plot Ip against √ν and get a straight line through the origin, your process is diffusion-controlled — a classic diagnostic for confirming a freely diffusing redox couple in solution.
- Slow scan rates (5–50 mV/s): More time for diffusion; well-resolved peaks; ideal for initial characterisation and formal potential measurement.
- Moderate scan rates (50–500 mV/s): Good for studying reaction kinetics and following up chemical reactions.
- Fast scan rates (>1 V/s): Used to characterise fast electron transfer and short-lived intermediates; require a potentiostat with a high-speed sampling rate.
A key diagnostic: if a peak current scales with scan rate (not √ν), your reaction is surface-confined — meaning the redox-active species is adsorbed onto the electrode rather than freely diffusing in solution. This is important for characterising modified electrodes, electrocatalysts, and biosensor surfaces.
6 Real-World Applications of Cyclic Voltammetry
Cyclic voltammetry is not a purely academic technique. It is used daily in research laboratories and industrial settings across Australia and the world to solve real problems. Here are the six most important application areas:
Battery & Energy Storage Research
CV characterises electrode materials for lithium-ion, sodium-ion, and redox flow batteries. Researchers use it to identify redox peaks, assess reversibility, estimate charge-storage behaviour, and screen new anode and cathode materials before committing to more extensive cell testing.
Biosensor Development
Biosensor researchers use CV to confirm that their modified electrode surface is functioning correctly, to measure the electrochemical signal of their target analyte, and to characterise the electron transfer between biological molecules and the electrode.
Corrosion Science
CV helps corrosion scientists understand the passivation behaviour of metals, map the potential regions where protective oxide films form and break down, and screen coatings and inhibitors for their protective effectiveness.
Electrocatalysis
CV is a primary screening technique for electrocatalysts for the hydrogen evolution reaction (HER), oxygen evolution reaction (OER), oxygen reduction reaction (ORR), and CO₂ reduction. It identifies the onset potential and activity of new catalyst materials.
Pharmaceutical & Drug Analysis
Many pharmaceutically active molecules are electrochemically active. CV is used to study drug oxidation mechanisms, screen for antioxidant activity, and develop electrochemical detection methods for drugs in biological matrices.
Nanomaterials Characterisation
CV characterises the electrochemical surface area of nanoparticles, confirms the successful functionalisation of nanostructured electrodes, and measures the specific capacitance of supercapacitor materials such as graphene and metal oxides.
7 Cyclic Voltammetry vs Other Electrochemical Techniques
CV is just one technique in a rich toolkit of electrochemical methods. Knowing when to use CV — and when to use something else — is an important part of experimental design.
| Technique | What It Does | Key Advantage over CV | Best Used When |
|---|---|---|---|
| CV | Sweeps voltage forward and backward; measures current at each point | — | New system, unknown chemistry; reversibility assessment |
| Linear Sweep Voltammetry (LSV) | Forward sweep only — no reverse | Faster; simpler data | Catalyst onset potential screening; one-directional reactions |
| Chronoamperometry (CA) | Fixed potential step; measures current decay over time | Quantitative kinetics at a single potential | Diffusion coefficients; sensor responses; fixed-potential kinetics |
| EIS | Sinusoidal voltage at multiple frequencies; measures impedance | Quantifies resistance, capacitance, diffusion parameters | Interface characterisation; batteries; corrosion; biosensors |
| DPV / SWV | Pulse-based voltammetry | Much lower detection limits | Trace-level quantitative detection |
When to Use Which
New system, unknown chemistry? Start with CV. Need kinetics or interface characterisation? Follow up with EIS. Quantitative detection at a fixed potential? Use CA or DPV. Catalyst onset potential screening? Use LSV. CV is the foundation from which all other decisions flow.
8 Which Potentiostat Do You Need for Cyclic Voltammetry?
The good news for anyone starting out with cyclic voltammetry is that CV is supported by virtually every potentiostat on the market — from compact, portable instruments costing a few thousand dollars to advanced research-grade platforms. The question is not can your potentiostat run CV? but rather which level of performance and additional capability does your research require?
At ScienceGears Australia, we supply potentiostats from three established manufacturers — Zahner Elektrik (Germany), Admiral Instruments (USA), and CorrTest Instruments — each covering a different research context and budget level.
Choose the Right CV Potentiostat for Your Research Stage. All instruments support cyclic voltammetry; additional capabilities (EIS, multichannel, high-current) vary by model. GST-inclusive pricing and Australian warranty support on all instruments.
- Students & Field Use
Portable Potentiostats
Techniques CV, LSV, CA, and more. Some models include EIS. View Portable Potentiostat Range →
- PhD Research
Single-Channel Potentiostats
Techniques Wide potential windows, low-noise current measurement, full EIS capability. View Single-Channel Potentiostat Range →
- Advanced Labs
Modular Zennium Systems
Techniques High-precision EIS, expandable channels, deep customisation. View Modular Range →
Step-by-Step: How to Choose
- Define your techniques first. CV only? An entry-level portable or single-channel will suffice. Need EIS alongside CV? Ensure your chosen model includes a frequency response analyser (FRA). Need galvanostatic experiments too? Many modern potentiostats include galvanostat mode.
- Consider your current range. Biosensor work with modified electrodes? You need pA–nA sensitivity. Battery materials at the gram scale? You may need mA–A range. Check the current range specifications carefully before purchasing.
- Think about your scan rate requirements. Standard CV work in the 10–500 mV/s range is commonly supported. Fast-scan CV can require a high-bandwidth potentiostat with fast sampling and control architecture.
- Request a quote and ask about Australian support. Ensure your supplier provides local warranty management, technical support in Australian time zones, and installation assistance. ScienceGears provides all of this from our Australian-based PhD team.
9 Frequently Asked Questions About Cyclic Voltammetry
These are the questions we receive most often from Australian researchers and lab managers before starting with cyclic voltammetry.
Q1 What is cyclic voltammetry used for?
Q2 Is cyclic voltammetry hard to learn?
Q3 What solvent and electrolyte should I use for CV?
Q4 What does a flat cyclic voltammogram mean?
Q5 Do I need EIS alongside cyclic voltammetry?
Q6 Can I run cyclic voltammetry on a portable potentiostat?
Q7 How much does a potentiostat for cyclic voltammetry cost in Australia?
Q8 What scan rate should I start with for a new CV experiment?
Q9 What is the difference between a two-electrode and three-electrode CV setup?
Q10 Why are my oxidation and reduction peaks not symmetric?
Q11 How do I choose the right reference electrode for aqueous vs non-aqueous CV?
Q12 Can cyclic voltammetry be used for battery materials before full charge-discharge testing?
Q13 When should I choose an EIS-capable potentiostat instead of a CV-only system?
Summary: Cyclic Voltammetry
Cyclic voltammetry is one of the most important tools in modern chemistry and materials science — and once you understand the fundamentals, it becomes an intuitive and powerful window into the electrochemical world. To summarise what we have covered in this guide:
- What CV is : A technique that sweeps voltage forward and backward across an electrode and measures the current at each point.
- What it produces : A cyclic voltammogram — a current vs potential graph showing oxidation and reduction peaks.
- What you need: A potentiostat, three electrodes (working, reference, counter), an electrolyte solution, and software.
- What the peaks tell you: The formal potential, reversibility of the reaction, peak current (linked to concentration), and kinetics of electron transfer.
- What it is used for : Batteries, biosensors, corrosion, catalysts, drugs, nanomaterials — and virtually any other electrochemical system you can place in a cell.
- Which potentiostat to choose : For students/field use, a portable potentiostat; for PhD research, a single-channel benchtop system with EIS.
If you are setting up a cyclic voltammetry capability for the first time — or upgrading your current system — the team at ScienceGears is here to help. Our co-founders, Dr. Siva Arumugam and Dr. Kalai Govindasamy, are active electrochemists with over 35 combined years of research experience. We do not just sell instruments — we help you use them effectively.
Research Behind This Guide
This guide was authored by Dr. Sivanesan Arumugam and Dr. Kalaivani Govindasamy, electrochemists with combined peer-reviewed publication records spanning biosensors, nanomaterials, and spectroelectrochemistry. Their published research directly informs the practical guidance in this article.
- Arumugam et al. (2015) — Protein biosensor using CV and EIS complementarity. Journal of Nanobiotechnology 13:43. (Open access — supports Section 7: CV vs EIS.)
- Arumugam et al. (2014) — SERS enhancement factor quantified via CV peak analysis. Electrochemistry Communications 49:103–106.
- Govindasamy et al. (2013) — Spectroelectrochemistry with CV validation (lead-author). RSC Advances 3(19):6839–6846.
- Full publication lists:Dr. Sivanesan Arumugam (Google Scholar) • Dr. Kalaivani Govindasamy (Google Scholar)