The MEA is where the electrochemical magic happens the compact, engineered “active sandwich” that decides whether your fuel cell delivers stable power or your electrolyser produces hydrogen efficiently at meaningful current density. In practice, most performance problems blamed on “the catalyst” or “the membrane” turn out to be interfaces: contact resistance, compression, sealing, flow distribution, and water/gas management.
This guide explains the MEA in clear, research-friendly terms, then connects the fundamentals to how MEAs are actually tested in the lab. You will learn the five critical components that control performance, where MEAs are used, how to test them, and how to choose the right test cell platform (graphite, metal, interchangeable flow field, visual, stack testing, and a turnkey package).ScienceGears MEA testing range is designed for PEM electrolysis (PEMWE),AEM electrolysis (alkaline environment),fuel cell development, and broader membrane electrochemistry.
What is a Membrane Electrode Assembly?
A membrane electrode assembly (MEA) is the functional core of many fuel cells and electrolysers: a layered structure where an ion-conducting membrane sits between catalyst-coated electrodes (often supplied as a catalyst-coated membrane, CCM) and, in many lab stacks, is paired with diffusion/transport media (GDL/PTL). In PEM systems, the membrane conducts protons while separating gases; in AEM systems, the membrane conducts anions (often hydroxide in water electrolysis, but carbonate/bicarbonate can dominate in CO₂-containing systems) and separates compartments. The MEA is where electrochemical reactions occur, while the surrounding hardware provides compression, flow distribution, and current collection.
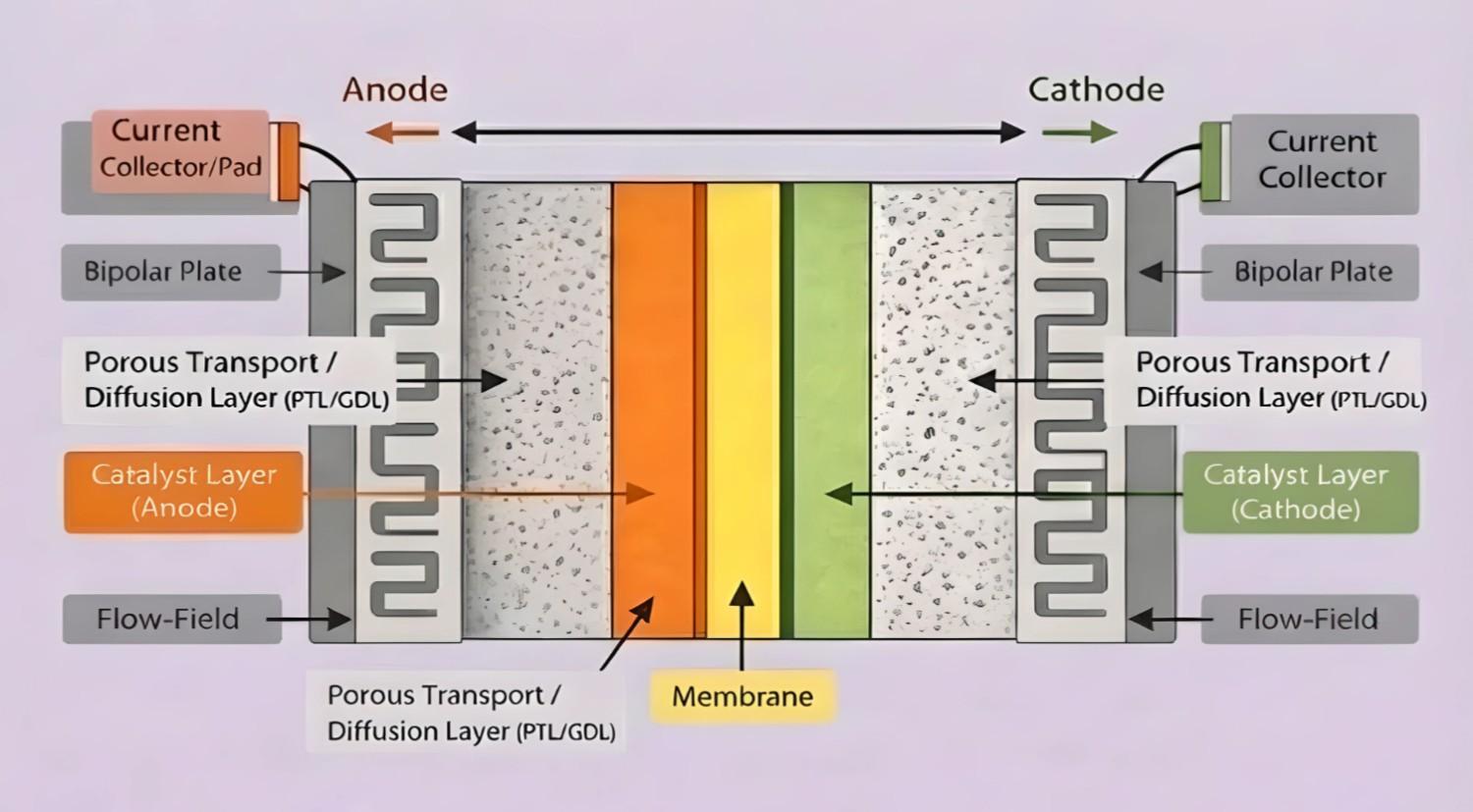
Schematic cross-section of a membrane electrode assembly (MEA) stack showing the anode and cathode sides, including current collectors, bipolar plates with serpentine flow-field channels, porous transport/diffusion layers (PTL/GDL), catalyst layers, and the central ion-exchange membrane; arrows indicate the direction of current flow across the cell.
The 5 critical components of an MEA
A practical way to think about MEA performance is: ions, electrons, gases and water must all move efficiently, at the same time, through thin layers. Any bottleneck (or leak) shows up as lost efficiency, instability, or faster degradation.
1) Ion-conducting membrane (PEM or AEM)
The membrane is the MEA’s ion highway and separator. It needs high ionic conductivity, low gas crossover, mechanical robustness, and chemical stability in the intended environment. In PEM devices, the membrane supports proton transport; in AEM devices, it supports anion transport. The membrane’s hydration state (and how your cell hardware manages it) strongly affects resistance and repeatability.
What researchers typically track:
- Ohmic resistance (often using EIS)
- Crossover/leak indicators and safety checks
- Mechanical integrity after cycling (edges, pinholes, creep)
2) Catalyst layers (anode and cathode)
Catalyst layers provide the active sites for charge transfer reactions. The “best” catalyst in a half-cell does not always win in an MEA because transport and interfacial contact matter: ionomer distribution, pore structure, and water management can dominate at realistic current density.
What researchers typically track:
- Activation losses (kinetic region)
- Stability during holds/cycles
- Sensitivity to impurities and contamination
3) Porous transport / diffusion media (PTL or GDL)
Diffusion/transport layers deliver reactants to the catalyst and remove products (gas and/or water). They also spread current and help maintain uniform compression. In the MEA context, this layer is part of the “sandwich” used in PEM fuel cells (diffusion media), and analogous transport layers are essential in electrolysis and membrane reactors.
Common performance symptoms tied to PTL/GDL:
- Flooding (transport limitation at higher currents)
- Dry-out / de-wetting (rising resistance, unstable voltage; most common in fuel cells and gas-fed systems)
- High contact resistance (persistent voltage penalty)
4) Gaskets and seals
Sealing is not just a mechanical detail; it is a reproducibility requirement. Leakage, bypass flow, and uneven compression can distort performance curves and make MEA comparisons meaningless.
What researchers typically control:
- Gasket thickness (compression window)
- Torque/fastening procedure
- Chemical compatibility with electrolytes and reactants
5) Current collectors + flow-field / bipolar plates
Even with a high-quality MEA, the surrounding plates often determine how well the device performs. Bipolar plates/flow fields help distribute reactants, conduct current between cells in a stack, support mechanical clamping, and contribute to water/heat management — which is why plate design and material choice are core research variables.
In short: the MEA is the electrochemical core, but plates/flow fields and compression decide whether the core can operate efficiently and repeatably.
MEA applications: where are they used?
Four major fields that use MEA-style (membrane + electrode) architectures or closely related membrane-flow electrochemical platforms
- Water electrolysis (PEMWE / AEMWE)– hydrogen generation, materials screening, and durability studies under controlled flow and compression
- Fuel cells (PEMFC and related)– MEA optimisation, gas diffusion layer (GDL) tuning, and water/heat management studies
- CO₂ electroreduction (CO₂RR) in membrane reactors / zero-gap cells– controlling cathode and anode environments while maintaining ionic conduction and managing flooding/carbonate formation
- Electrochemical separations (related membrane + flow systems) – ion removal using electrically driven separation concepts (e.g., electrodialysis (ED), and MCDI/CDI variants; these are adjacent to, not classic, PEM/AEM MEA devices)
ScienceGears’ MEA test cell platform family supports controlled MEA testing for research and prototyping. Start here:
Application requirements vs MEA design focus (quick comparison)
| Application area | What matters most | MEA & hardware focus | Typical lab measurements |
|---|---|---|---|
| PEM/AEM water electrolysis (PEMWE/AEMWE) | Efficiency + durability (stable voltage at high current, long-life operation) | Sealing and stable compression, PTL/GDL selection, catalyst–ionomer layer quality, low contact resistance, corrosion/coating stability, flow-field distribution, gas/liquid management | Polarisation curves, EIS / HFR, long holds (steady-state), durability cycling, leak checks, gas crossover / purity checks |
| Fuel cell testing (e.g., PEMFC) | Power density + water management (avoid flooding/dry-out while maintaining performance) | Diffusion media (GDL/MPL) tuning, humidification/water balance, flow-field design, compression uniformity, contact resistance control, temperature management | Polarisation curves, EIS / HFR, durability cycling, (optional diagnostics: CV/LSV depending on protocol) |
| CO₂RR membrane reactors / zero-gap CO₂ cells | Selectivity (Faradaic efficiency) + stable transport (high partial current density without flooding/salt issues) | Flow-field and gas delivery control, GDE/wetting behaviour, bubble and water management, carbonate/salt precipitation control, stable compression and sealing, membrane choice (AEM/CEM/BPM as required) | Product analysis (FE/selectivity) + electrochemistry (cell voltage, j–V), stability runs, (often: outlet gas/liquid quantification over time) |
| Related: electrochemical separations (ED / MCDI / CDI) | Salt removal performance + energy efficiency | Electrode architecture, membrane/ion-exchange interfaces (where applicable), controlled flow and residence time, minimising pressure drop, stable electrical contact | Conductivity/salt removal vs time, charge passed, charge efficiency, energy per volume (or per mole removed), cycle stability |
How to test an MEA (procedure overview)
This is a general testing guide that works with many different MEA test setups. You can use it for electrolysis, membrane reactor experiments, and fuel cell–type tests. The aim is to test in a consistent way so your results are repeatable and easy to compare fairly.
- Define your objective: Catalyst screening? Membrane comparison? Flow-field optimisation? Long-duration durability?
- Choose the test cell platform: Select a fixture or package matched to your objective (single cell, visual diagnostics, flow-field studies, or stack testing).
- Document the full stack-up: Record membrane type, electrode/catalyst layer details, transport layers, gasket thickness, and active area.
- Assemble with controlled compression: Use a consistent torque sequence. Compression affects contact resistance, leakage, and transport behaviour.
- Condition the MEA (as per your protocol): Conditioning stabilises interfaces and hydration state before “real” data collection.
- Collect baseline performance + diagnostics: Polarisation curve, EIS, and leak/crossover checks appropriate to your setup.
- Run stability testing: Use holds or cycles that match your research goal (short screening vs long-duration degradation).
- Post-test inspection: Look for gasket imprint patterns, flooding traces, edge damage, delamination hints, and transport layer deformation.
Key metric to understand: Current density (A/cm²) normalises current by active area, enabling fair comparisons between different MEA sizes and flow-field geometries.
Choosing the right MEA test cell platform (with ScienceGears product links)
This is where education and practical buying guidance overlap: the “best” platform depends on what variable you are trying to control.
Graphite Bipolar Plate MEA Electrolysis Test Cell (general single-cell testing)
Designed for reproducible compression and sealing, and described as accommodating PEM or AEM membranes for studies including CO₂RR and AEM water electrolysis (alkaline environment).
View product page : Graphite Bipolar Plate MEA Electrolysis Test Cell
Best for:
- Baseline single-cell MEA experiments
- Fast comparative screening with repeatable assembly
Metal Bipolar Plate MEA Electrolysis Test Cell (Ti/Ni/SS) (temperature-controlled, robust)
Built around high-purity metal plates (typically titanium) with features such as double O-ring sealing and ports for heating/temperature measurement, enabling tighter control for AEM water electrolysis (alkaline environment), PEM studies, and CO₂RR.
View product page : Metal Bipolar Plate MEA Electrolysis Test Cell (Ti/Ni/SS)
Best for:
- Temperature-controlled testing and sustained operation
- Experiments where mechanical strength and conductivity matter
Interchangeable Channel MEA Electrolytic Cell (flow-field experimentation)
Designed for rapid flow-field studies, using corrosion-resistant plates and interchangeable channel geometries (commonly serpentine) to study mass transport and flooding behaviour under otherwise identical conditions.
View product page : Interchangeable Channel MEA Electrolytic Cell
Best for:
- “Is it chemistry or transport?” investigations
- Optimising flow distribution, pressure drop, bubble removal, and flooding resistance
Visual MEA Electrolysis Test Cell (diagnostics and in-situ observation)
Uses optical windows for in-situ observation/high-speed imaging, aimed at diagnosing gas evolution, flooding, and channel utilisation. Supports studies of flow-field distribution effects.
View product page : Visual MEA Electrolysis Test Cell
Best for:
- Troubleshooting instability, flooding, or bubble release issues
- Visual validation of channel utilisation and distribution hypotheses
MEA Electrolysis Cell Stack (stack/scale-up studies)
A stackable architecture designed to increase test voltage/power by combining multiple flow-plate units, with ports for heating/temperature measurement and configurable flow fields. Positioned for prototyping, durability testing, and scale-up studies.
View product page : MEA Electrolysis Cell Stack
Best for:
- Cell-to-cell repeatability studies
- Scale-up and long-duration protocols where single-cell results are not enough
Turnkey MEA Complete Package (all components included)
A complete package intended to help labs get to data faster by bundling the MEA test cell with key peripherals such as a gas separator, electrolyte reservoir, peristaltic pump, heating/temperature control, and integration tubing/fittings.
View product page : Turnkey MEA Complete Package
Best for:
- New MEA labs setting up capability quickly
- Teams that want a single-source, compatible kit for immediate experiments
Bonus sidebar: Bipolar plate vs bipolar membrane (don’t mix these up)
These two terms sound similar, and they cause real confusion — but they are fundamentally different.
Bipolar plate
A bipolar plate is a hardware component in many MEA devices (fuel cells and some electrolysers) It helps distribute reactants through flow fields, conducts current between cells in a stack, supports mechanical clamping, and contributes to water/heat management.
Bipolar membrane (BPM)
A bipolar membrane is a type of ion-exchange membrane constructed from an anion-exchange layer and a cation-exchange layer. At their interface, water dissociation can generate H⁺ and OH⁻ under a reverse-bias electric field (often aided by a catalytic junction layer) — which is why BPMs are used in certain electrolysis, CO₂RR, and separations concepts.
Key takeaway: Your product pages refer to bipolar plates (hardware/flow fields). A bipolar membrane is a specific membrane chemistry and structure.
Common MEA challenges and practical solutions
1) Flooding / bubble build-up and mass-transport limits
Symptoms: instability or rapid performance change at higher current density; strong sensitivity to flow rate; poor repeatability.
What helps: improve flow distribution, manage bubbles/water, and (if possible) observe behaviour directly.
Useful platforms:
2) Poor hydration (dry-out) and rising ohmic resistance (system-dependent)
Symptoms: increasing HFR/ohmic loss; unstable operation; worsening EIS ohmic component.
What helps: stabilise wetting/hydration, ensure even compression, and tune flow-field and operating conditions.
Note: In liquid-fed electrolysis, classic dry-out is less common; bubble management and local de-wetting are more typical contributors to rising resistance and instability.
3) High contact resistance (compression/contact issues)
Symptoms: uniformly worse voltage across the whole polarisation curve; high HFR (EIS/current interrupt).
What helps: repeatable torque pattern, clean/flat interfaces, robust collectors/plates, and stable temperature control.
Related platform: Metal Bipolar Plate MEA Electrolysis Test Cell
4) Leakage and sealing failures (external leaks or internal crossover)
Symptoms: gas crossover, unstable readings, safety concerns, inconsistent performance (often worse under differential pressure).
What helps: correct gasket design/material, torque discipline, pressure/leak checks, and sealing-focused cell designs (e.g., O-ring features where applicable).
5) “Single cell works, stack fails” (scale-up effects)
Symptoms: strong single-cell results, but stack testing shows cell-to-cell variation or instability.
What helps: stackable architecture with controlled compression, good manifold design/flow distribution, thermal management, and long-duration repeatable assembly protocols.
6) Interface/catalyst-layer mechanical failure (delamination/cracking/pinholes)
Symptoms: gradual or sudden performance loss, rising HFR, abnormal EIS, failure after cycling.
What helps: avoid over-compression, consistent assembly, appropriate conditioning, and post-test inspection to identify root cause.
7) Chemical degradation / contamination (materials compatibility)
Symptoms: drift over time, unexpected losses, rising resistance, inconsistent repeats across builds.
What helps: high-purity inputs, compatible materials/coatings, avoid corrosion sources, and clean handling.
Quick comparison table: which platform should you choose?
| Platform | Best for | Why it’s useful |
|---|---|---|
| Graphite bipolar plate MEA cell | General single-cell MEA testing | Reproducible compression/sealing; PEM/AEM compatible |
| Metal bipolar plate MEA cell (Ti/Ni/SS) | Temperature-controlled and robust testing | Heating/temperature measurement ports; strong sealing options |
| Interchangeable channel MEA cell | Flow-field optimisation | Swap channel geometries; designed for transport studies |
| Visual MEA cell | Diagnosing flooding/bubbles | Optical windows for in-situ observation |
| MEA electrolysis cell stack | Scale-up & durability | Stackable architecture; ports for temperature monitoring; prototyping |
| Turnkey MEA complete package | Fast setup | Includes cell + key peripherals (pump, reservoir, gas separator, heating control) |
Related technology (kept short): Capacitive Deionisation (CDI)
CDI is not a classic PEM/AEM “MEA device” in the strict fuel cell/electrolyser sense, but many labs working on electrochemical water treatment and separations use related electrode-and-flow architectures. If your team is extending membrane electrochemistry toward desalination or ion removal workflows, this category is a useful adjacent pathway:
Capacitive Deionisation (CDI) Cells
Conclusion (and a practical next step)
A membrane electrode assembly is the electrochemical core — but credible results depend on the full system: membrane, catalyst layers, transport media, sealing, compression, and flow-field design. If you want publishable MEA comparisons, prioritise repeatability (torque, gaskets, assembly method) and use the test platform that matches your objective: baseline screening, flow-field optimisation, visual diagnostics, or stack-level durability.
To compare ScienceGears’ MEA test cell fixtures and packages in one place, start here:
FAQs
1) What is the difference between an MEA and an MEA test cell?
An MEA is the membrane + catalyst-coated electrodes (often supplied as a CCM), and in many lab stacks the term is also used loosely to include the paired transport layers (GDL/PTL). An MEA test cell is the hardware fixture that compresses the MEA, seals it, routes fluids/gases through flow fields, and collects current reliably. Most reproducibility issues come from the test cell hardware and assembly conditions, not the MEA chemistry alone.
2) Can the same test cell hardware be used for both PEM and AEM membranes?
Often yes (hardware-wise) — many fixtures can physically accommodate either membrane type. But you must still match:
- Catalyst/ionomer chemistry(PEM vs AEM ionomer is not interchangeable)
- Operating environment(water/alkaline, CO₂RR conditions, etc.)
- Materials compatibility(especially metals in alkaline or CO₂ environments)
A safe statement is: “One fixture may support PEM or AEM configurations, but the MEA materials and operating protocol must match the membrane chemistry.”
3) Can an electrolyser be run “in reverse” as a fuel cell?
In principle, the core electrochemistry is reversible (water ↔ H₂ + O₂), but a typical electrolyser is not automatically a fuel cell in practice. The same hardware may not be optimised for:
- Gas delivery vs liquid handling
- Water management/humidification
- Catalyst choices (OER vs ORR catalysts are different)
- Safety and balance-of-plant requirementsSo: “Reversible operation exists conceptually, but most lab electrolysers require changes to MEA, flow setup, and operating conditions to run as a fuel cell.”
4) What is “HFR” and why do MEA researchers care about it?
HFR (High Frequency Resistance) is a practical estimate of the cell’s high-frequency ohmic resistance (typically dominated by the membrane and interfacial/contact resistances, and sometimes including small fixture/lead contributions). If HFR is high, your performance drops everywhere (not just at high current). Tracking HFR helps separate:
- Ohmic losses(membrane/contact/compression)from
- Kineticand mass transport limitations
5) Why does torque/compression matter so much?
Compression controls:
- Contact resistance(plates ↔ PTL/GDL ↔ catalyst)
- Sealing(leaks/crossover)
- Porosity and transport(over-compression can reduce pore volume/permeability and increase two-phase transport losses such as flooding or bubble blockage). For publishable comparisons, you need a repeatable torque pattern and gasket thickness.
6) What causes flooding in MEA testing, and how do I diagnose it fast?
Common causes include:
- Flow-field geometry not suited to your gas/liquid regime
- Too much liquid retention in transport layers
- Uneven compression or local blockageBest diagnostics:
- Compare performance at different flow rates
- Use interchangeable flow fieldsto isolate transport effects
- Use a visual cellto literally confirm bubble build-up / channel utilisation
7) What is the benefit of an interchangeable flow-field MEA cell?
It lets you test whether performance is limited by:
- chemistry (catalyst/membrane)or
- transport (flow distribution, bubble removal, pressure drop) By holding everything else constant and swapping channels, you can defend mechanistic claims much more strongly.
8) When do I need a visual MEA test cell?
Use a visual cell when you are troubleshooting:
- Unstable voltage
- Sudden performance drop at higher current
- Suspected bubble blockage, flooding, maldistribution, or uneven wettingIt’s also helpful for developing newflow-field designs because you can validate assumptions quickly.
9) Graphite bipolar plates vs metal bipolar plates — when should I choose each?
A practical (non-controversial) way to frame it:
- Graphite-based setupsare widely used for general MEA experiments and can be a good baseline platform.
- Metal plate setups (e.g., titanium) are often chosen when you want robustness, higher mechanical strength, or tighter temperature/control features, and when materials compatibility with your chemistry is appropriate.(Keep it as “selection depends on chemistry and protocol,” which is defensible.)
10) What active area should I choose for MEA testing?
For early-stage R&D, many teams start smaller to:
- reduce MEA cost
- accelerate screening
- improve controlAs you scale active area, you learn about distribution, sealing, and repeatability— which is where stack testing becomes valuable.
11) Why do single-cell results fail to translate to stack performance?
Stacks introduce:
- Cell-to-cell variation in compression/contact
- Manifold and flow distribution effects
- Thermal gradients and water management differencesThat’s why a stack test fixtureis essential once you move beyond screening.
12) What does a “turnkey MEA test package” save time on?
It reduces setup risk and time by bundling compatible components (typical examples are):
- Reservoir/recirculation hardware
- Gas/liquid separation and fittings
- Temperature/heating control integrationThis is most useful when a lab wants to start generating data immediatelywithout designing balance-of-plant from scratch.
13) What are the most common “MEA test mistakes” that ruin data quality?
Top repeatability killers:
- Changing gasket thickness without recording it
- Inconsistent torque sequence
- Poorly controlled conditioning protocol
- Leaks/crossover not checked
- Using the wrong ionomer/binder for PEM vs AEM
14) Is CDI an MEA technology?
Not in the strict PEMFC/PEMWE sense. CDI is a related electrochemical separation technology that shares “electrode + flow + ion transport” concepts. It belongs as a short “related technology” mention (as you requested), not a main MEA section.







